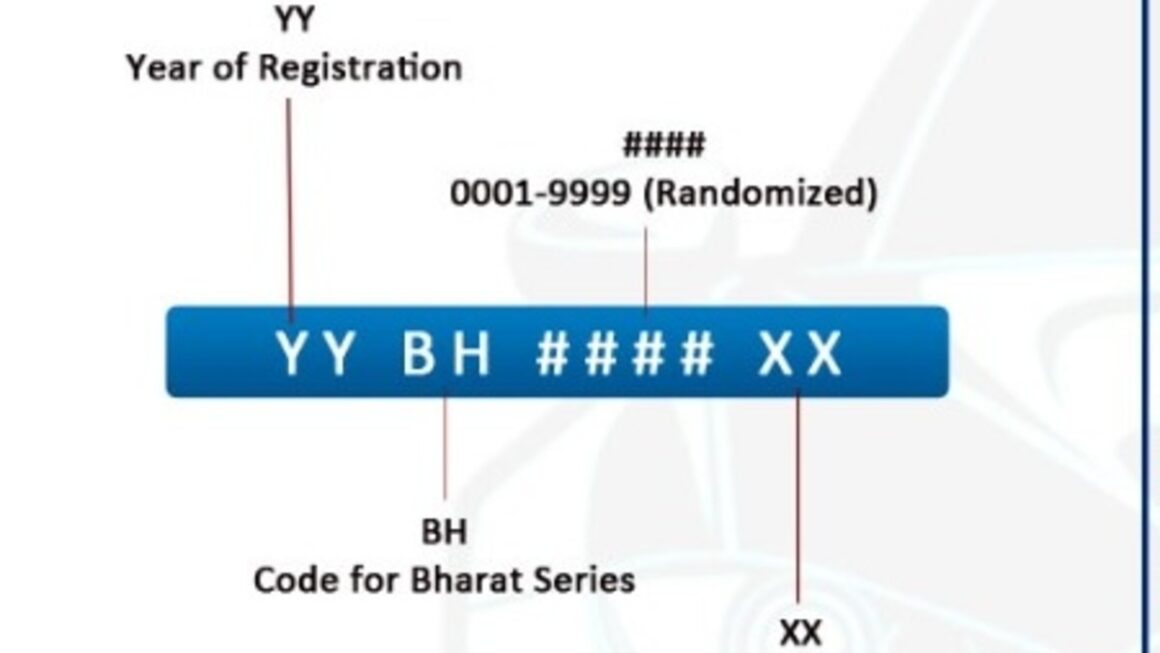
SOLVED: Consider a weak base equilibrium as shown in the equation below: What is the effect on Kb if BH were added to the solution? B (aq) + H2O () = BHt (
![SOLVED: The base B has pKb 5.00. (a) What is the value of pKa for the acid BH? (b) At what pH is [BH] [B]? (c) Which is the principal species, B SOLVED: The base B has pKb 5.00. (a) What is the value of pKa for the acid BH? (b) At what pH is [BH] [B]? (c) Which is the principal species, B](https://cdn.numerade.com/ask_previews/fddc0959-fd50-4e65-bc4a-81b32be9fdcd_large.jpg)
SOLVED: The base B has pKb 5.00. (a) What is the value of pKa for the acid BH? (b) At what pH is [BH] [B]? (c) Which is the principal species, B