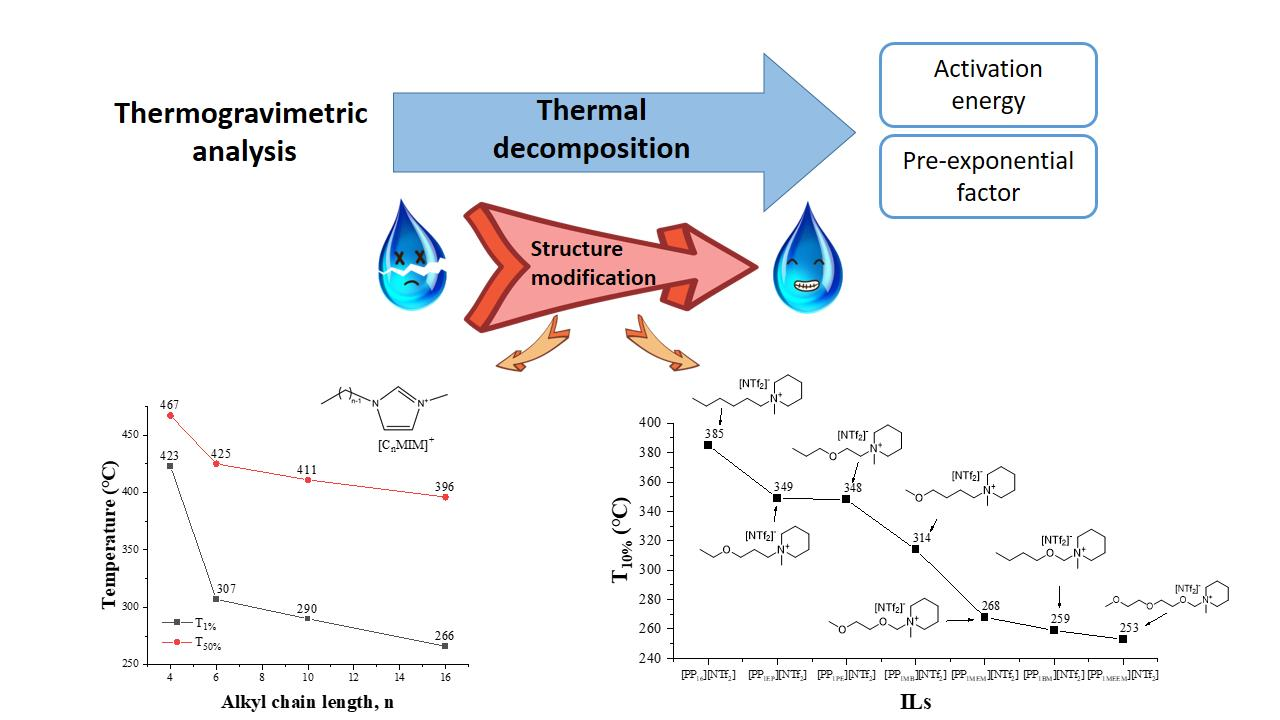
Processes | Free Full-Text | Thermal Stability of Ionic Liquids: Current Status and Prospects for Future Development

Thermal analysis of palladium (II) acetate (a) in argon; (b) in air.... | Download Scientific Diagram
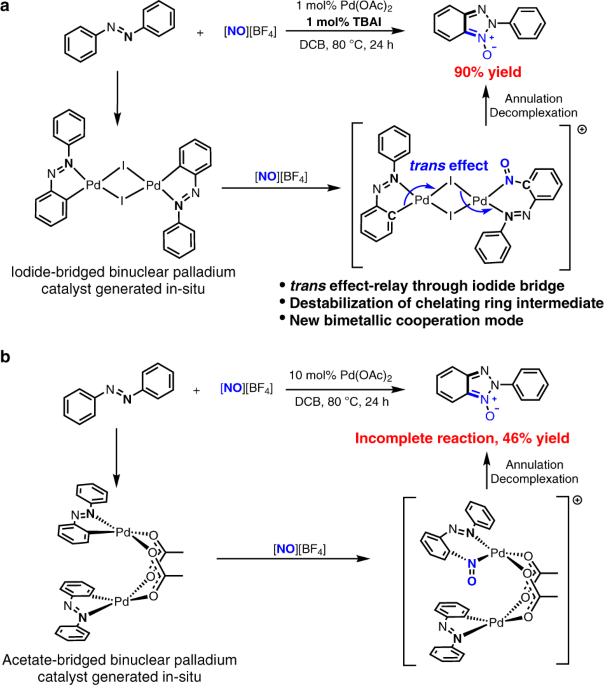
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry

Room-Temperature Phosphorescence from Pd(II) and Pt(II) Complexes as Supramolecular Luminophores: The Role of Self-Assembly, Metal–Metal Interactions, Spin–Orbit Coupling, and Ligand-Field Splitting | Journal of the American Chemical Society
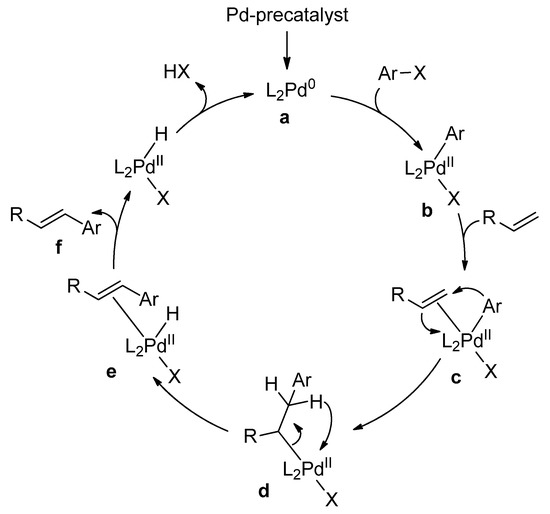
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond
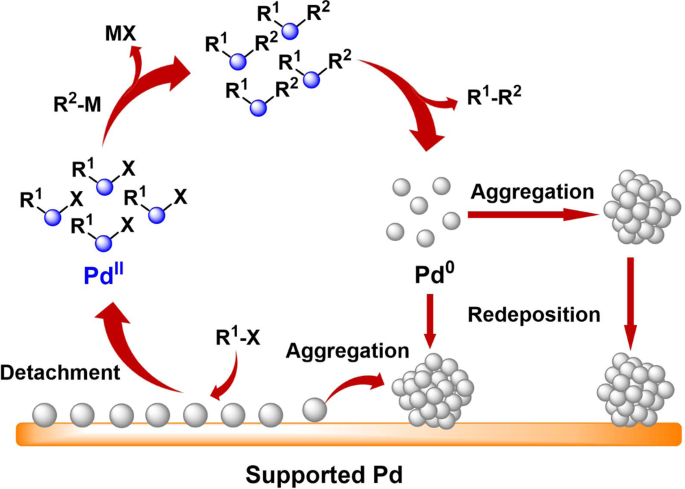
Enhancing stability by trapping palladium inside N-heterocyclic carbene-functionalized hypercrosslinked polymers for heterogeneous C-C bond formations | Nature Communications

Catalysts | Free Full-Text | Catalytic Performance and Reaction Mechanisms of Ethyl Acetate Oxidation over the Au–Pd/TiO2 Catalysts

The Palladium Acetate‐Catalyzed Microwave‐Assisted Hirao Reaction without an Added Phosphorus Ligand as a “Green” Protocol: A Quantum Chemical Study on the Mechanism - Keglevich - 2017 - Advanced Synthesis & Catalysis -

Cationic Palladium(II) Catalysis: C−H Activation/Suzuki−Miyaura Couplings at Room Temperature | Journal of the American Chemical Society

On the Mechanism of Palladium-Catalyzed Aromatic C−H Oxidation | Journal of the American Chemical Society

Palladium Acetate Trimer: Understanding Its Ligand-Induced Dissociation Thermochemistry Using Isothermal Titration Calorimetry, X-ray Absorption Fine Structure, and 31P Nuclear Magnetic Resonance | Organometallics

Decarbonylative Fluoroalkylation at Palladium(II): From Fundamental Organometallic Studies to Catalysis | Journal of the American Chemical Society

Processes | Free Full-Text | Thermal Stability of Ionic Liquids: Current Status and Prospects for Future Development