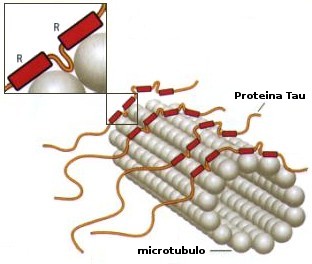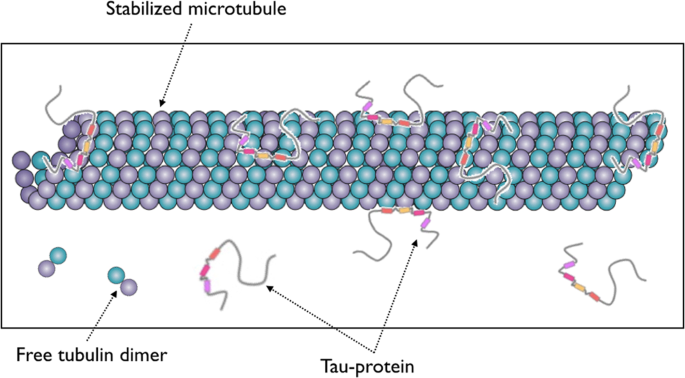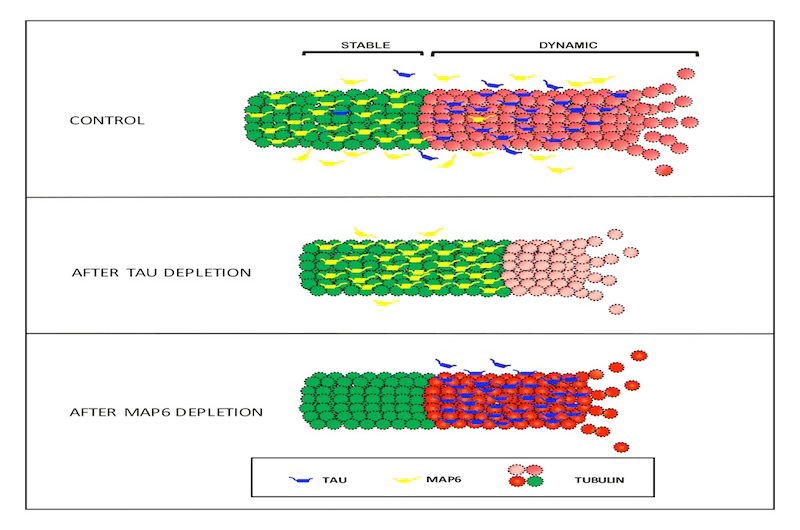
Study Finds Tau Protein Does Not Stabilize Microtubules, Challenges Approach to Treating Alzheimer's

Site-specific hyperphosphorylation of tau inhibits its fibrillization in vitro, blocks its seeding capacity in cells, and disrupts its microtubule binding; Implications for the native state stabilization of tau | bioRxiv
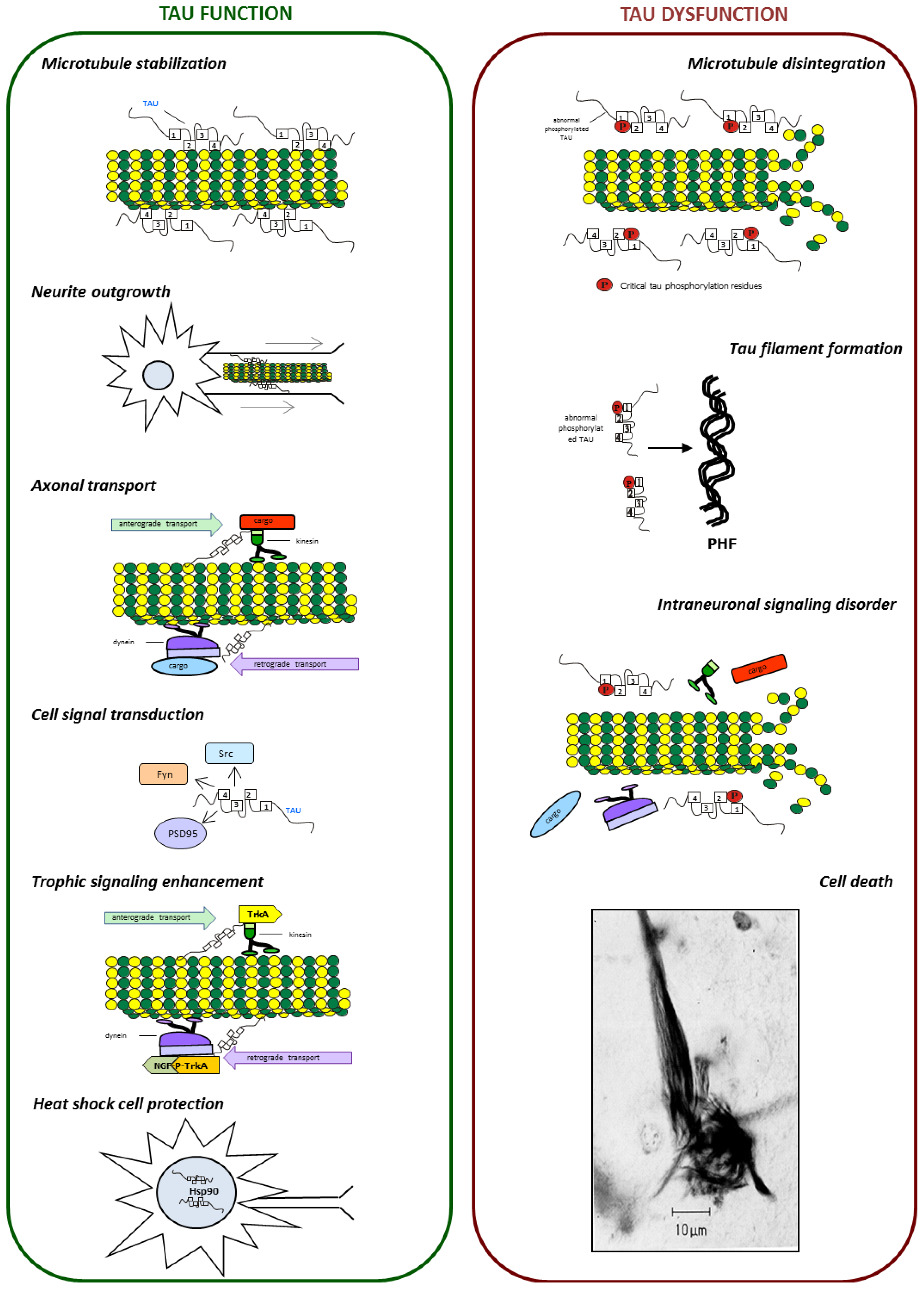
IJMS | Free Full-Text | Tau Protein Modifications and Interactions: Their Role in Function and Dysfunction

Oligomerization of the microtubule‐associated protein tau is mediated by its N‐terminal sequences: implications for normal and pathological tau action - Feinstein - 2016 - Journal of Neurochemistry - Wiley Online Library

Minireview - Microtubules and Tubulin Oligomers: Shape Transitions and Assembly by Intrinsically Disordered Protein Tau and Cationic Biomolecules | Langmuir

Tau protein. (A) In physiological conditions, tau binds to microtubules... | Download Scientific Diagram
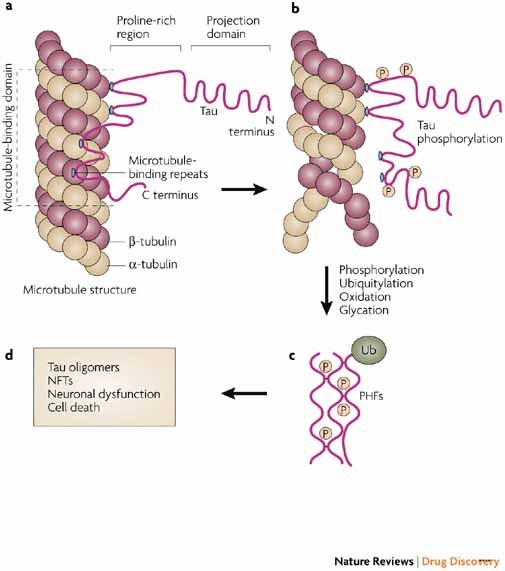
Untangling tau hyperphosphorylation in drug design for neurodegenerative diseases | Nature Reviews Drug Discovery

Advances in tau-focused drug discovery for Alzheimer's disease and related tauopathies. - Abstract - Europe PMC
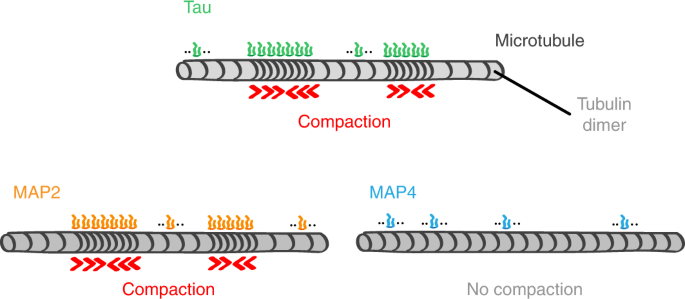
Microtubule lattice spacing governs cohesive envelope formation of tau family proteins | Nature Chemical Biology
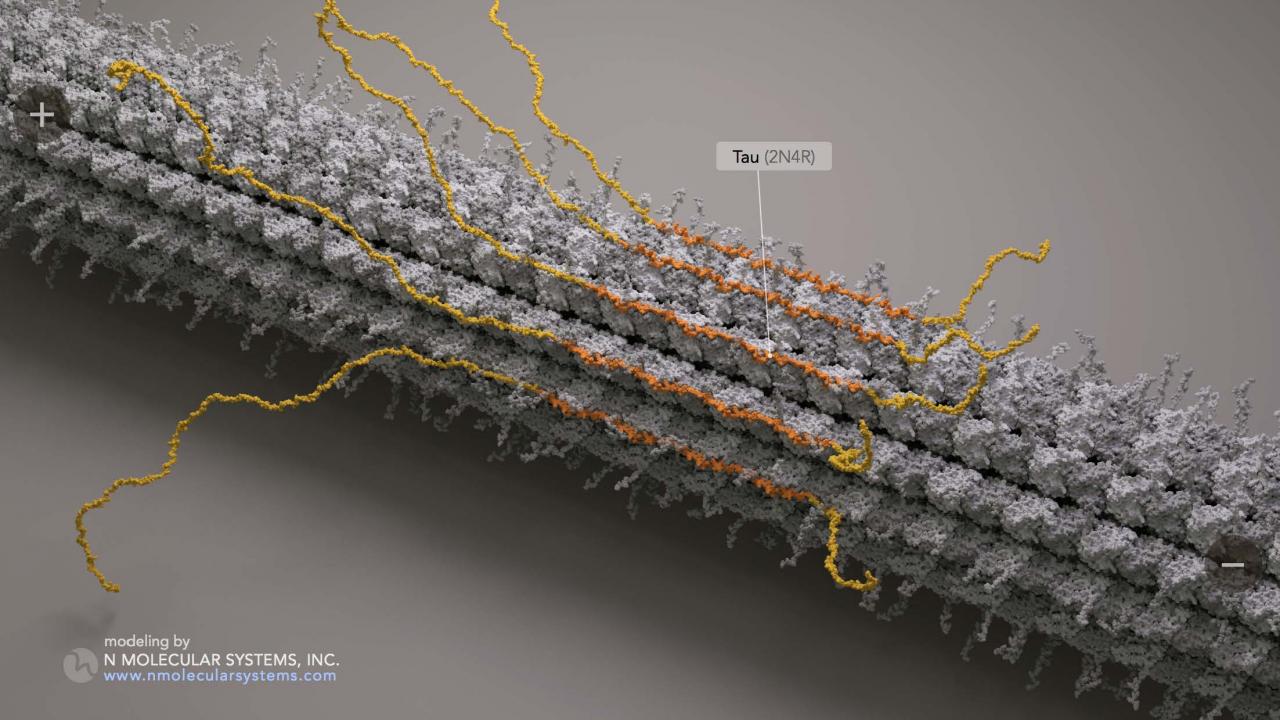
Gatekeeper of the Cellular Highway: Study Reveals Novel Behaviors of the Alzheimer's Disease Protein Tau | College of Biological Sciences
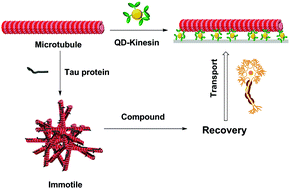
Modulating the microtubule–tau interactions in biomotility systems by altering the chemical environment - Integrative Biology (RSC Publishing)


![PDF] Conformation of Human Microtubule Associated Protein-Tau | Semantic Scholar PDF] Conformation of Human Microtubule Associated Protein-Tau | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/74b85c7c51f3b65a903a99cc0e97e5c642579c9c/8-Figure1.2-1.png)