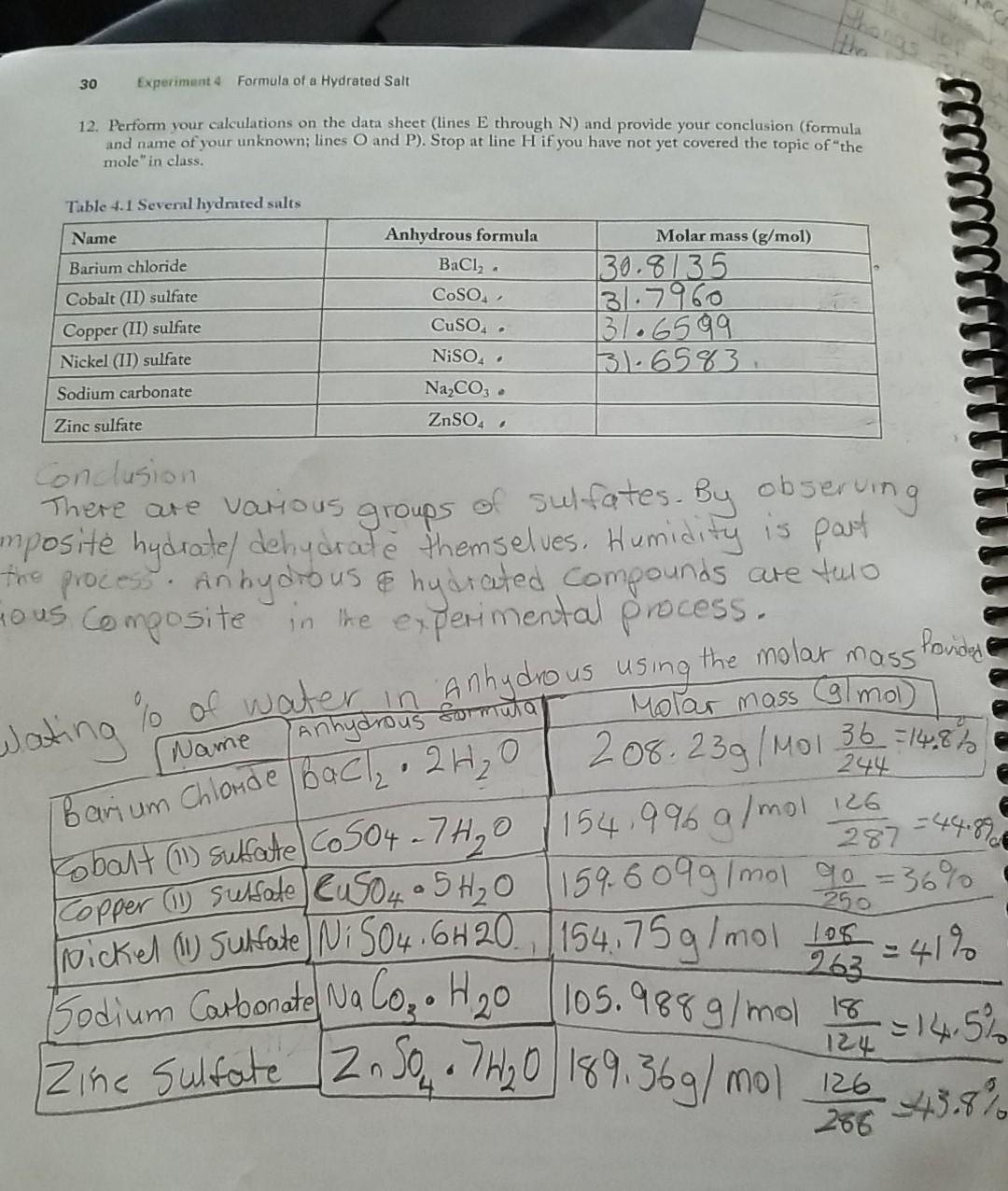
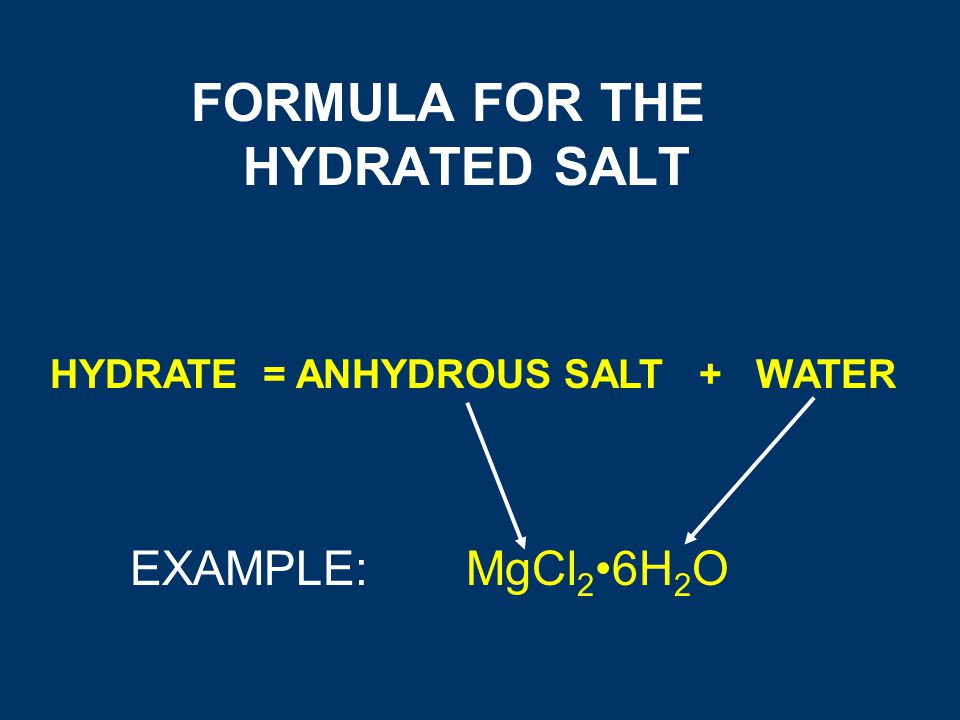
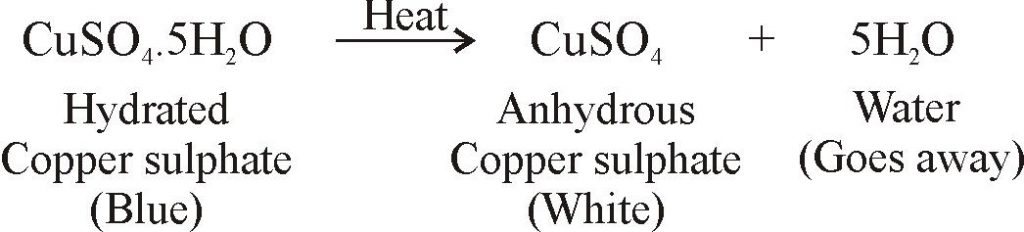
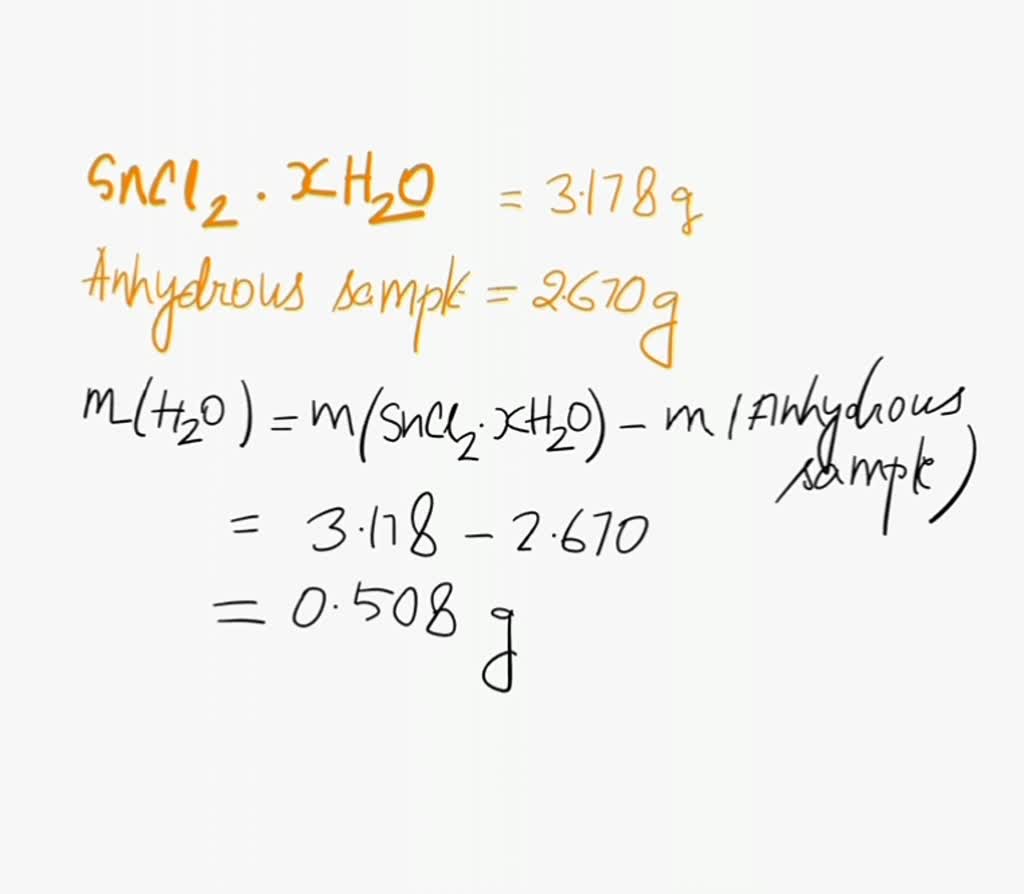Welcome to Chem Zipper.com......: The hydrated salt, Na2SO4.nH2O undergoes 55.9% loss in weight on heating and becomes anhydrous. The value of n will be?

Question Video: Calculating the Percentage of Water of Crystallization Given the Mass of the Hydrated and Dehydrated Salt | Nagwa

State of the art on salt hydrate thermochemical energy storage systems for use in building applications - ScienceDirect

A hydrated salt is a solid that includes water molecules within its crystal structure. A student - Brainly.com