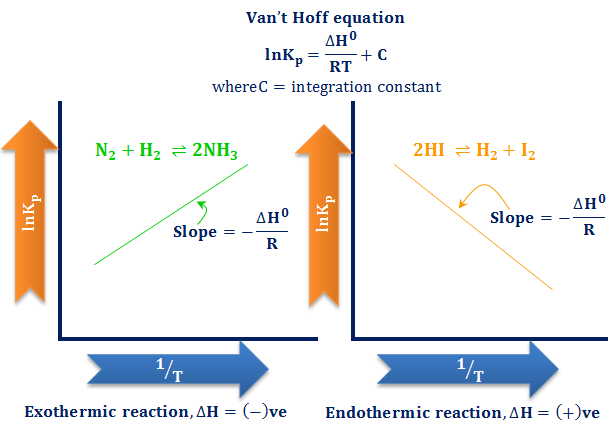
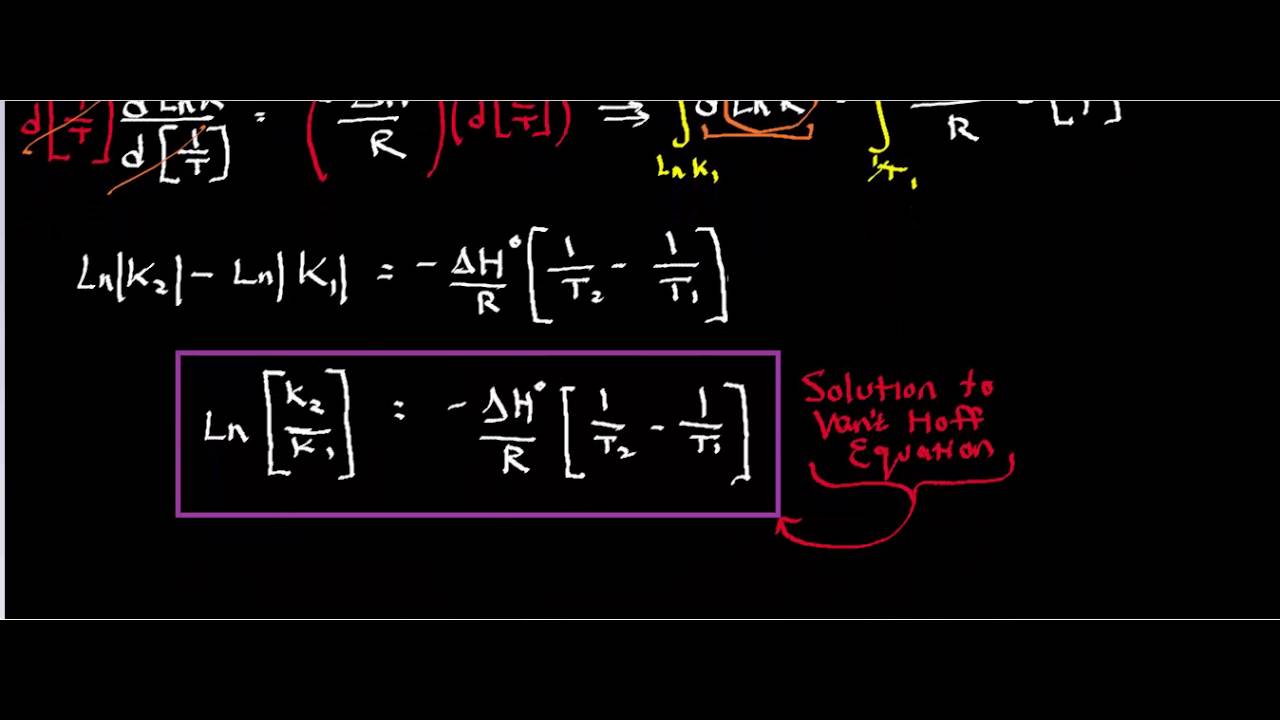
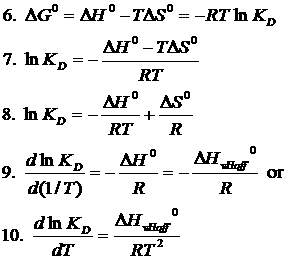van't Hoff's equation gives the quantitative relation between change in value of K with change in temperature.

PDF) Van't Hoff equation-equilibrium How much Van't Hoff equation -effect on temperature | Kajal Panda - Academia.edu

vant hoff equation for osmotic pressure of solution and expiration of degree of dissolution - Chemistry - - 14038313 | Meritnation.com